Lactate Fermentation
In lactate fermentation, pyruvate is converted to lactate by the enzyme lactate dehydrogenase. In the process, NAD+ is regenerated. The reaction catalyzed by lactate dehydrogenase (LDH) is shown in Figure (PageIndex{2}).
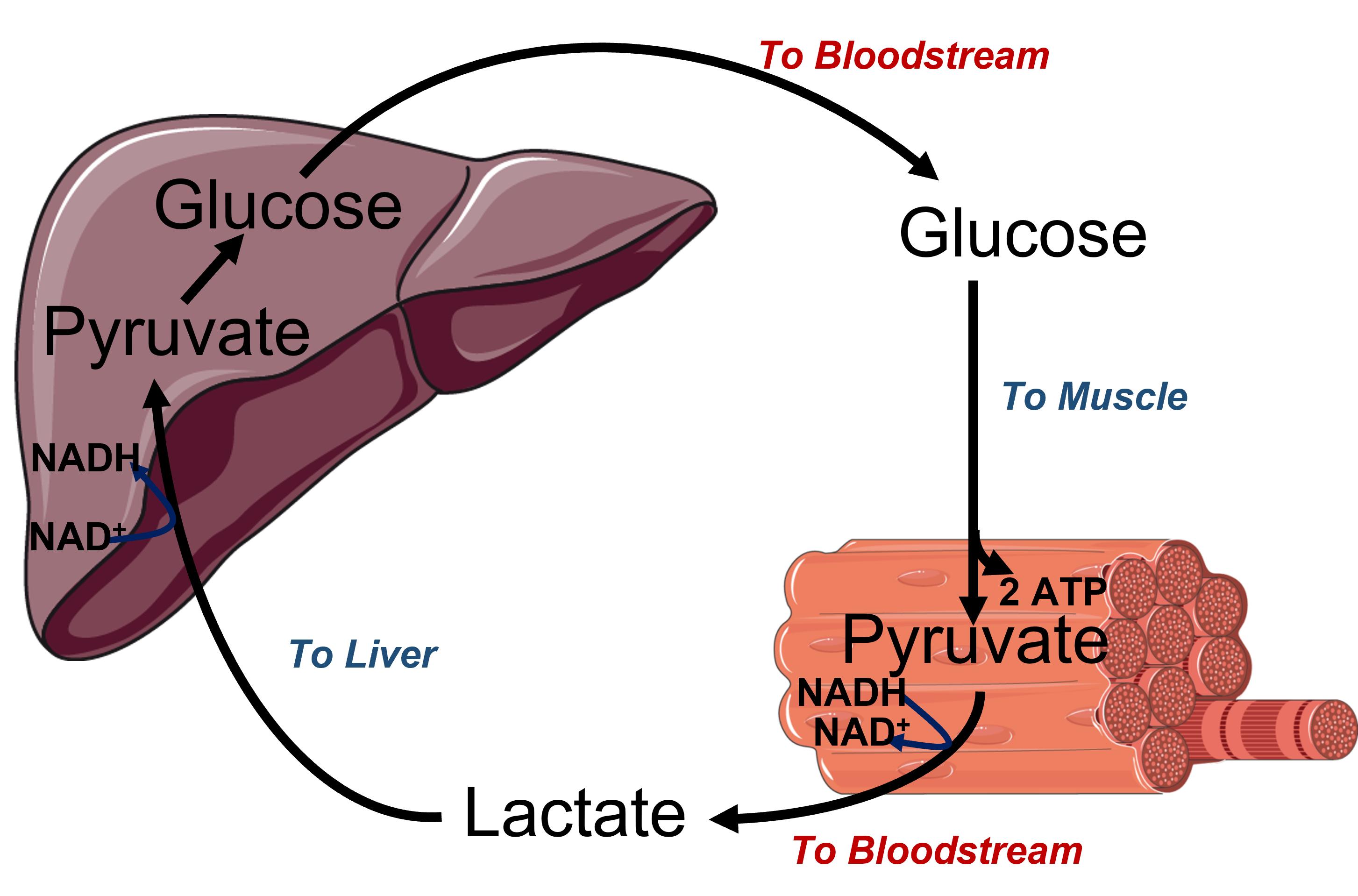
Figure (PageIndex{2}): Reaction catalyzed by lactate dehydrogenase
It is named for the reverse reaction which, as with other dehydrogenases, uses NAD+ as an oxidizing agent. The reaction is reversible with the ΔG0 for conversion of pyruvate to lactate of -3.76 kcal/mol (-15.7 kJ/mol). Under anaerobic conditions when glycolysis is the major source of ATP, pyruvate levels increase, further driving the reaction towards lactate formation and NAD+ regeneration so glycolysis can continue.
The enzyme is found in the cytoplasm but a mitochondrial form also exists. It is most abundant in muscle, liver, kidney, and also in erythrocytes. Interestingly, mature red blood cells are enucleated and do not contain any mitochondria. Their lifespan is limited to approximately two weeks. During this time, their primary energy resources are generated through the process of anaerobic fermentation via the glycolysis-lactate pathway.
The active enzyme is a tetramer of various compositions of two different subunits, the heart (H) and muscle (M) forms. The quaternary structures consist of 5 different isozyme forms containing the H and M subunits. LDH-1, found most abundantly in the heart, is a tetramer of 4H subunits (H4). The other forms are as follows: LDH-2 (H3M, prevalent in RBCs), LDL-3 (H2M2, prevalent in lungs), LDH-4 (HM3, prevalent in the kidney), and LDH-5 (M4, prevalent in muscle).
During anaerobic metabolism, lactate is produced by muscle tissue and released into the bloodstream where it can travel back to the liver. Once in the liver, lactate is converted back into pyruvate and can be utilized to produce glucose through a pathway called gluconeogenesis. The liver can then export the glucose into the blood from where it can be taken up by the muscle for ATP production. This cycle is called the Cori cycle and is illustrated in Figure (PageIndex{3}).
.png)
.png)
Figure (PageIndex{3}): Cori cycle Upper diagram is a cartoon image of the Cori cycle and the lower diagram demonstrates the recycling of NAD+/NADH in different locations within the body. Figure modified from Servier Medical Art
Notably, anaerobic metabolism can only be sustained for short periods in animals due to its high energy demand. Aerobic respiration is required to maintain an adequate ATP supply. However, the production of lactate by certain tissues, such as white, or fast-twitch muscle cells occurs regularly and releases lactate into the bloodstream, where it can be taken up and used as an energy source by other neighboring tissues such as red, slow-twitch muscle. Brain tissue can also effectively use lactate as an energy source as well. Within these ‘consumer’ tissues, lactate is converted back into pyruvate using the mitochondrial LDH enzyme where it can then be processed by aerobic respiration producing high levels of ATP.
Thus, the production of lactate can be thought of as a strain response that occurs during times of metabolic stress, such as intense cardiovascular exercise. It has also been noted that after an injury or head trauma, the activation of epinephrine will cause an increase in lactate production and blood levels of lactate will increase. Thus, it has been hypothesized that lactate may play a role in the repair of damaged tissue. Clinical trials and experiments are currently underway to determine if lactate can help in the healing and recovery process for conditions such as traumatic brain injury, myocardial infarction, and sepsis. Early studies have shown that lactate can increase the production of Brain-Derived Neurotropic Factor (BDNF) which supports neuronal growth, providing further support for the role of lactate in recovery and repair.
Spectroscopy analyses show the geometry and electrostatics of the active sites of LDHs from all organisms are essentially the same. The mechanism for the reverse oxidation of lactate by NAD+ by spiny dogfish lactate dehydrogenase (LDH) is shown in Figure (PageIndex{4})
Figure (PageIndex{4}): Mechanism for the reduction of lactate by NAD+ by spiny dogfish lactate dehydrogenase (LDH)
Read more : Why Does My Jaw Hurt When I Yawn
Lactate is shown with a deuterium (D), which moves as a deuteride to NAD+ to form NADH, simply to illustrate the stereochemistry of the reaction. In the reverse reaction, the reduction of pyruvate to lactate (to regenerate more NAD+ so anaerobic glycolysis can continue), the deuterium (or the proR H of non-deuterated NADH) is removed.
Figure (PageIndex{5}) shows an interactive iCn3D model of dogfish M4 apo-lactate dehydrogenase (1LDM).
Figure (PageIndex{5}): Dogfish M4 apo-lactate dehydrogenase (1LDM).(Copyright; author via source). Click the image for a popup or use this external link: https://structure.ncbi.nlm.nih.gov/i…FBzuqJ2gEPUHM8
Only the monomer of the active tetramer is shown. NAD and oxamic acid, structurally similar to lactate/pyruvate and a competitive inhibitor of the enzyme, are labeled. His193, the active site general base/acid, is near Arg 169 and to Asp 166 and Asn 138, to which it is hydrogen bonded.
LDH exists in two major conformational states, T (inactive) and R (active) state, as we have seen with other allosteric proteins. Figure (PageIndex{6}) shows an interactive iCn3D model of the T state (2ZQY) and R state (2ZQZ) of Lacticaseibacillus casei L-lactate dehydrogenase.
Figure (PageIndex{6}): T state (2ZQY) and R state (2ZQZ) of Lacticaseibacillus casei L-lactate dehydrogenase. (Copyright; author via source). Click the image for a popup or use this external link: https://structure.ncbi.nlm.nih.gov/i…zjrtVYjLXdhDp8
Toggle between the magenta T state and cyan R state using the “a” key. Orient the proteins to see the best view of the allosteric changes caused by binding regulators. The spheres show Arg 171 whose disposition changes significantly in the T and R states. Pyruvate (a reactant/product) activates the enzyme (conversion of T to R state) and is considered a homotropic regulator since it is a substrate. This is similar to the allosteric “activation” of hemoglobin by its ligand O2, which preferentially binds to the R state and promotes the T-to-R state transition. Fructose 1,6-bisphosphate, a glycolytic intermediate, also activates the Lacticaseibacillus casei L-lactate dehydrogenase and since it is not a reactant/product of the enzyme, it is considered a heterotropic allosteric regulator. The enzyme hence is effectively regulated by the concentration of substrate (pyruvate) and by NADH levels. High concentrations of ethanol consumption lead to high levels of NADH through the activity of alcohol dehydrogenase. High levels of NADH would lead to increased lactate production as well.
Source: https://t-tees.com
Category: WHY



